COBLATION – Sticks for Adenotonsillectomy
A range of sticks for tissue ablation, resection, and coagulation* in various procedures using WEREWOLF COBLATION Technology – proven to operate at lower temperatures compared to other methods**¹–⁴

A range of sticks for tissue ablation, resection, and coagulation* in various procedures using WEREWOLF COBLATION Technology – proven to operate at lower temperatures compared to other methods**¹–⁴
Thin-edged cutting tip, additional rear electrode for coagulation, and the most durable electrode in our ENT wand portfolio, designed to help minimize blockage.⁵
Proven to provide access to the choanae during adenoidectomy, with a flexible shaft up to 6 inches and triple-wire electrode.⁶,⁷
Equipped with the same flexible shaft and triple-wire active electrode, shown to enable efficient tissue removal (in vivo).⁸
Large, flat-screen electrode has been shown to provide more than 20% faster ablation compared to the EVAC 70 XTRA Wand (ex vivo).⁹
Comparable performance to the EVAC 70 XTRA Wand, but with a lower-profile shaft that may be better suited for smaller anatomies.⁹–¹¹
Contains a small electrode and has been shown in lab testing to provide fine dissection.¹²
Combines ablation, suction, and bipolar hemostasis in a compact design, providing precision and visibility during surgery.¹³–¹⁶
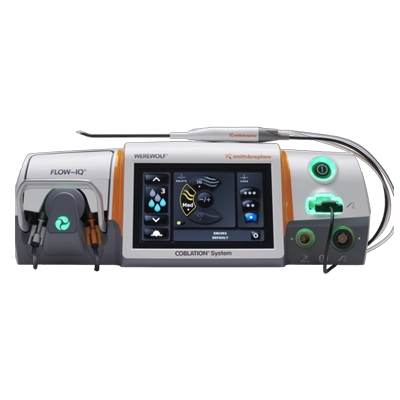
COBLATION Technology provides precise tissue ablation while minimizing thermal damage to surrounding tissues.¹⁷,¹⁸
Operates at lower temperatures compared to other ablation methods for adenotonsillectomy procedures (typically not exceeding 40–65°C). **¹–⁴,¹⁷,¹⁸
Unlike electrocautery or CO2 laser surgery, laboratory tests have shown that COBLATION Technology reduces the risk of airway fire.¹⁹–²¹
Has been used in tonsil and adenoid surgeries for at least 20 years and is supported by over 70 peer-reviewed publications.²²,²³
1. Smith+Nephew 2010. Internal report. PN 69832 Rev A.
2. Smith+Nephew 2010. Internal report. PN 86791 Rev 1.
3. Smith+Nephew 2010. Internal report. P/N 86257 Rev A.
4. Smith+Nephew 2010. Internal report. P/N 60736-01 Rev A.
5. Smith+Nephew 2020. Internal report. P/N 113227 Rev A.
6. Mularczyk C, et al. Int J Pediatr Otorhinolaryngol. 2018;104:29 – 31.
7. Smith+Nephew 2004. Internal report. P/N 12595 Rev C.
8. Smith+Nephew 2010. Internal report. PN 69832 Rev A.
9. Smith+Nephew 2010. Internal report. P/N 37785-01_A.
10. Smith+Nephew 2011. Internal report. 35684-01 Rev A.
11. Smith+Nephew 2007. Internal report. 18752 Rev A.
12. Smith+Nephew. Internal report. 14409 Rev B.
13. Varadharajan K, et al. Int J Pediatr Otorhinolaryngol. 2020;135:110113.
14. Hoey AW, et al. Clin Otolaryngol. 2017;42(6):1211 – 1217.
15. Smith+Nephew 2007. Internal report. 21480 Rev A.
16. Smith+Nephew 2008. Internal report. 21487.
17. Magdy EA, et al. J Laryngol Otol. 2008;122(3):282 – 290.
18. Roje Z, et al. Coll Antropol. 2009;33(1):293 – 298.
19. Roy S, et al. Am J Otolaryngol – Head Neck Med Surg. 2010;31(5):356 – 359.
20. Roy S, et al. Am J Otolaryngol – Head Neck Med Surg. 2015;36(1):63 – 66.
21. Matt BH, et al. Otolaryngol – Head Neck Surg. 2010;143(3):454 – 455.
22. Smith+Nephew 2017. Internal report. P/N 91999.
23. Temple RH, et al. Int J Pediatr Otorhinolaryngol. 2001;61(3):195 – 198.
24. Francis DO, et al. Otolaryngol Head Neck Surg. 2017;156(3):442-455.
25. Smith+Nephew 2021. Internal report. EA/ENT/COBLATION/002/V4.
Unlike total tonsillectomy, which removes all tonsil tissue including the capsular plane to expose larger blood vessels in the extracapsular area, intracapsular tonsillectomy aims to remove tonsil tissue by ablating up to the tonsil capsule.¹⁴
It has been shown to result in less pain and a lower risk of postoperative bleeding compared to total tonsillectomy.²⁴,²⁵
*Features depend on the selected wand. Please refer to the Instructions for Use (IFU) for each product.
**COBLATION ENT Technology operates at lower temperatures than monopolar RF technologies, laser technologies, and PEAK plasma blade.
For detailed product information, including indications, contraindications, precautions, and warnings, refer to the relevant Instructions for Use (IFU) before use.
Products may not be available in all markets, as availability depends on local regulations and/or medical practices. If you have questions about the availability of Smith+Nephew products in your region, please contact your Smith+Nephew representative or distributor.
◊Smith+Nephew is a trademark of Smith+Nephew. All trademarks acknowledged. ©2022 Smith+Nephew. All rights reserved.
Lara Group is a medical technology company operating in the field of medical technologies, aiming to add value to the healthcare sector through innovative solutions. Since our establishment, we have adopted a human-centered approach, bringing advanced medical products together with healthcare professionals.
Demirci Mh. Ahıska Cd.
Pratik Kaynak Merkezi
No.195/1 İç Kapı 1
Nilüfer/BURSA Türkiye
T.+90 532 375 98 35
info@laragroup.com.tr
Mo-Fr: 8:00-19:00
Sat: 8:00-14:00
Sun: Kapalı
 RAPID RHINO SINU-FOAM – SMITH+NEPHEW
RAPID RHINO SINU-FOAM – SMITH+NEPHEW